Your cart is currently empty!
Linoleic Acid
Role of Linoleic Acid in Stratum Corneum Health and Ceramide Production
Linoleic acid is an essential fatty acid that plays a vital role in skin health. It is a precursor to ceramides, which are lipids that are essential for the formation of the stratum corneum, the outermost layer of the skin. The stratum corneum acts as a barrier to protect the skin from environmental stressors and prevent trans-epidermal water loss (TEWL). The lipids that constitute the extracellular matrix of the stratum corneum have a unique composition and are 50% ceramides, 25% cholesterol, and 15% free fatty acids(1). A deficiency of Linoleic acid can lead to not only insufficient Ceramide production(2), but also to all the other symptoms that are associated with essential fatty acid (EFA) deficiency which include reduced barrier function and other skin ailments(3–7). The following text outlines the mechanism through which linoleic acid when applied to the skin in the source of a plant oil is metabolized through the actions of enzymes present in the epidermis to the form of a free fatty acid. Being in the form of a free fatty acid it is then available to be further processed to longer chain fatty acids that have biological functions in themselves or can be used as building blocks for ceramides.
Fatty Acids and Triglycerides
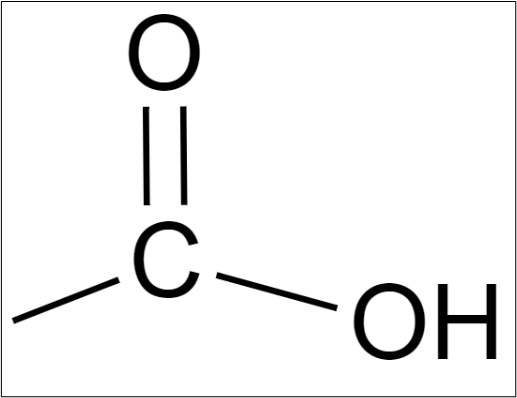
A fatty acid consists of a chain of carbon atoms, to which hydrogen atoms are bonded along the length of the chain and at one end of the chain a carboxyl group is found. It is that carboxyl group that makes it an acid. It is from the carboxyl group that this class of acids gets it name… carboxylic acid.
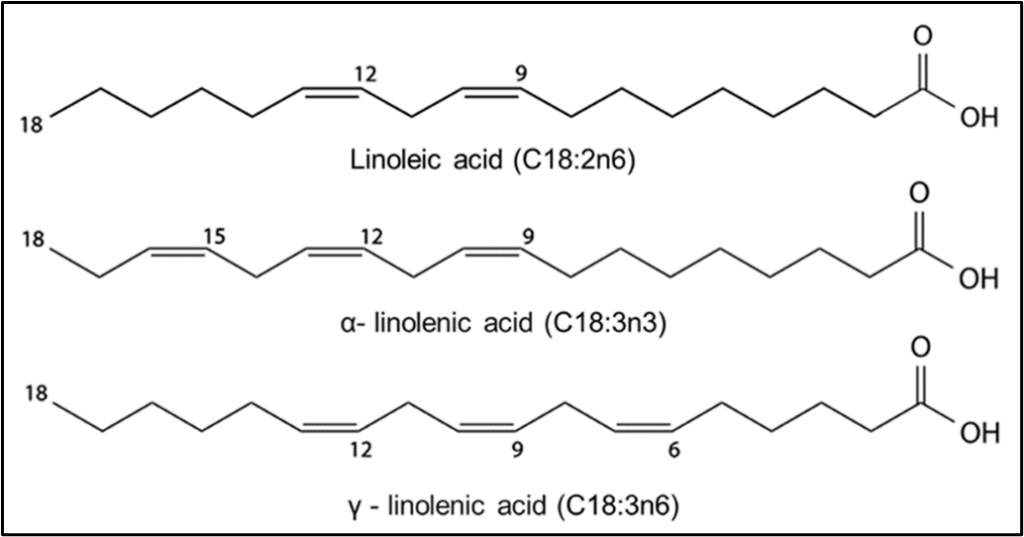
Below in Fig. 2 is a picture of three fatty acids, linoleic acid, alpha linolenic acid and gamma linolenic acid. In this picture the 18 carbon atom chain can be seen with double bonds along its length at various positions and a carboxyl group to the right end of the chain. (8)
In plant oils the fatty acids are bonded with a glycerin molecule to form a Triglyceride. A triglyceride is three fatty acids that are bonded to a glycerin molecule.
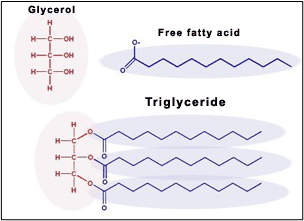
It is in the form of a Triglyceride that the fatty acids are introduced to the epidermis in the topical application of a cream that has plant oils as ingredients.
Metabolism of plant oils in the stratum corneum
In order for the cells of the epidermis to be able to use Linoleic acid and other fatty acids as building blocks for their metabolites the triglyceride must be broken down into its basic components of fatty acids and glycerin. The process by which a triglyceride is broken downinto its component fatty acids is called lipolysis. Lipolysis is a multi-step process that is catalyzed by a number of enzymes. The lipolytic cleavage of TGs occurs in essentially all cells and tissues of the body. In all of them, the resulting fatty acids are utilized endogenously for energy production or biosynthetic pathways(10).
The first step in lipolysis is the removal of one fatty acid from the triglyceride molecule. This is done by the enzyme Adipose Triglyceride Lipase (ATGL). The results of this action is a diacylglyceride molecule and a free fatty acid.
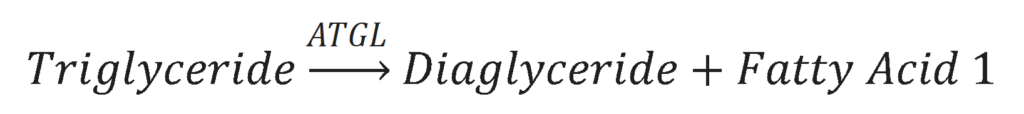
The second step in lipolysis is the removal of another fatty acid from the diacylglycerol molecule. This is done by the enzyme Hormone-Sensitive Lipase (HSL). The results of this reaction is a monoacylglycerol molecule and a second free fatty acid.
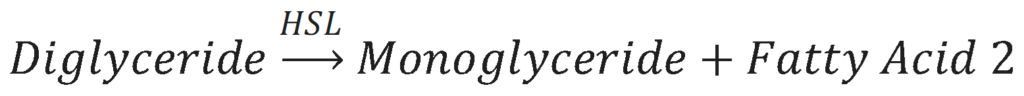
The third and final step in lipolysis is the removal of the last fatty acid from the monoglyceride molecule. This is done by the enzyme Monoglyceride Lipase (MGL). The results of this reaction is a glycerol molecule and a third free fatty acid.
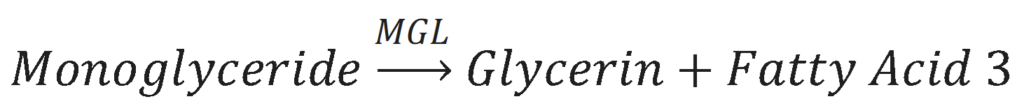
All three of the enzymes that catalyze lipolysis are present in the epidermis. Lipolysis is therefore able to occur in the epidermis, which allows the fatty acids that are released from triglycerides to be used by the epidermal cells.
Free fatty acids from triglycerides as building blocks for further Metabolites
Both linoleic acid and alpha linolenic acid are essential fatty acids meaning that the body can not make them itself and they need to be taken in from outside sources (11,12). The free fatty acids can be metabolized in the endoplasmic reticulum of cells into longer chain fatty acids by the enzymes present there (13–15). The metabolites that result either have a biological function in themselves or can be used as building blocks for even more complex molecules such as Sphingolipids and ceramides (16–19).

References
1. Feingold KR, Elias PM. Role of lipids in the formation and maintenance of the cutaneous permeability barrier. Biochim Biophys Acta BBA – Mol Cell Biol Lipids. 1. März 2014;1841(3):280–94.
2. Costa A, Siqueira Talarico A, Parra Duarte C de O, Silva Pereira C, de Souza Weimann ET, Sabino de Matos L, u. a. Evaluation of the Quantitative and Qualitative Alterations in the Fatty Acid Contents of the Sebum of Patients with Inflammatory Acne during Treatment with Systemic Lymecycline and/or Oral Fatty Acid Supplementation. Dermatol Res Pract. 2013;2013:120475.
3. Skolnik P, Eaglstein WH, Ziboh VA. Human essential fatty acid deficiency: treatment by topical application of linoleic acid. Arch Dermatol. Juli 1977;113(7):939–41.
4. Yamamoto A, Serizawa S, Ito M, Sato Y. Stratum corneum lipid abnormalities in atopic dermatitis. Arch Dermatol Res. 1. April 1991;283(4):219–23.
5. Truchetet E, Brändle I, Grosshans E. Skin changes, pathophysiology and therapy in deficiency of essential fatty acids. Z Hautkr. 15. April 1988;63(4):290–301.
6. Prottey C, Hartop PJ, Press M. Correction of the Cutaneous Manifestations of Essential Fatty Acid Deficiency in Man by Application of Sunflower-Seed Oil to the Skin. J Invest Dermatol. 1. April 1975;64(4):228–34.
1
7. Jeppesen PB, Høy CE, Mortensen PB. Essential fatty acid deficiency in patients receiving home parenteral nutrition12. Am J Clin Nutr. 1. Juli 1998;68(1):126–33.
8. Islam MS, Christopher LP, Alam MN. Separation and Purification of ω-6 Linoleic Acid from Crude Tall Oil. Separations. März 2020;7(1):9.
9. de Alaiz A. What are Triglycerides? 2018 [zitiert 11. Oktober 2023]. clinidiabet.com > cardiodiabetes > What are Triglycerides? Verfügbar unter: https://clinidiabet.com/en/infodiabetes/cardiodiabetes/15.htm
10. Schweiger M, Eichmann TO, Taschler U, Zimmermann R, Zechner R, Lass A. Measurement of Lipolysis. Methods Enzymol. 2014;538:171–93.
11. Yuan Q, Xie F, Huang W, Hu M, Yan Q, Chen Z, u. a. The review of alpha-linolenic acid: Sources, metabolism, and pharmacology. Phytother Res. 2022;36(1):164–88.
12. Mogensen KM. Essential Fatty Acid Deficiency. Pract Gastroenterol. 2017;
13. Rabionet M, Gorgas K, Sandhoff R. Ceramide synthesis in the epidermis. Biochim Biophys Acta. März 2014;1841(3):422–34.
14. Jakobsson A, Westerberg R, Jacobsson A. Fatty acid elongases in mammals: their regulation and roles in metabolism. Prog Lipid Res. Mai 2006;45(3):237–49.
15. Moon YA, Horton JD. Identification of two mammalian reductases involved in the two-carbon fatty acyl elongation cascade. J Biol Chem. 28. Februar 2003;278(9):7335–43.
16. Gault C, Obeid L, Hannun Y. An overview of sphingolipid metabolism: from synthesis to breakdown. Adv Exp Med Biol. 2010;688:1–23.
17. Cha HJ, He C, Zhao H, Dong Y, An IS, An S. Intercellular and intracellular functions of ceramides and their metabolites in skin (Review). Int J Mol Med. 1. Juli 2016;38(1):16–22.
18. Murff HJ, Edwards TL. Endogenous Production of Long-Chain Polyunsaturated Fatty Acids and Metabolic Disease Risk. Curr Cardiovasc Risk Rep. 1. Dezember 2014;8(12):418.
19. Blaess M, Wenzel F, Csuk R, Deigner HP. Topical use of amitriptyline and linoleic acid to restore ceramide rheostat in atopic dermatitis lesions – a case report. Pharm. 1. September 2019;74(9):563–5.
20. Leng GC, Taylor GS, Lee AJ, Fowkes FG, Horrobin D. Essential fatty acids and cardiovascular disease: the Edinburgh Artery Study. Vasc Med Lond Engl. 1999;4(4):219–26.
